List of oncology demo protocols
You will see the full view of each record. As in view mode "complete". Further details about the different viewscan be found in the license description on onkopti.de.
These protocols are intended for healthcare professionals.
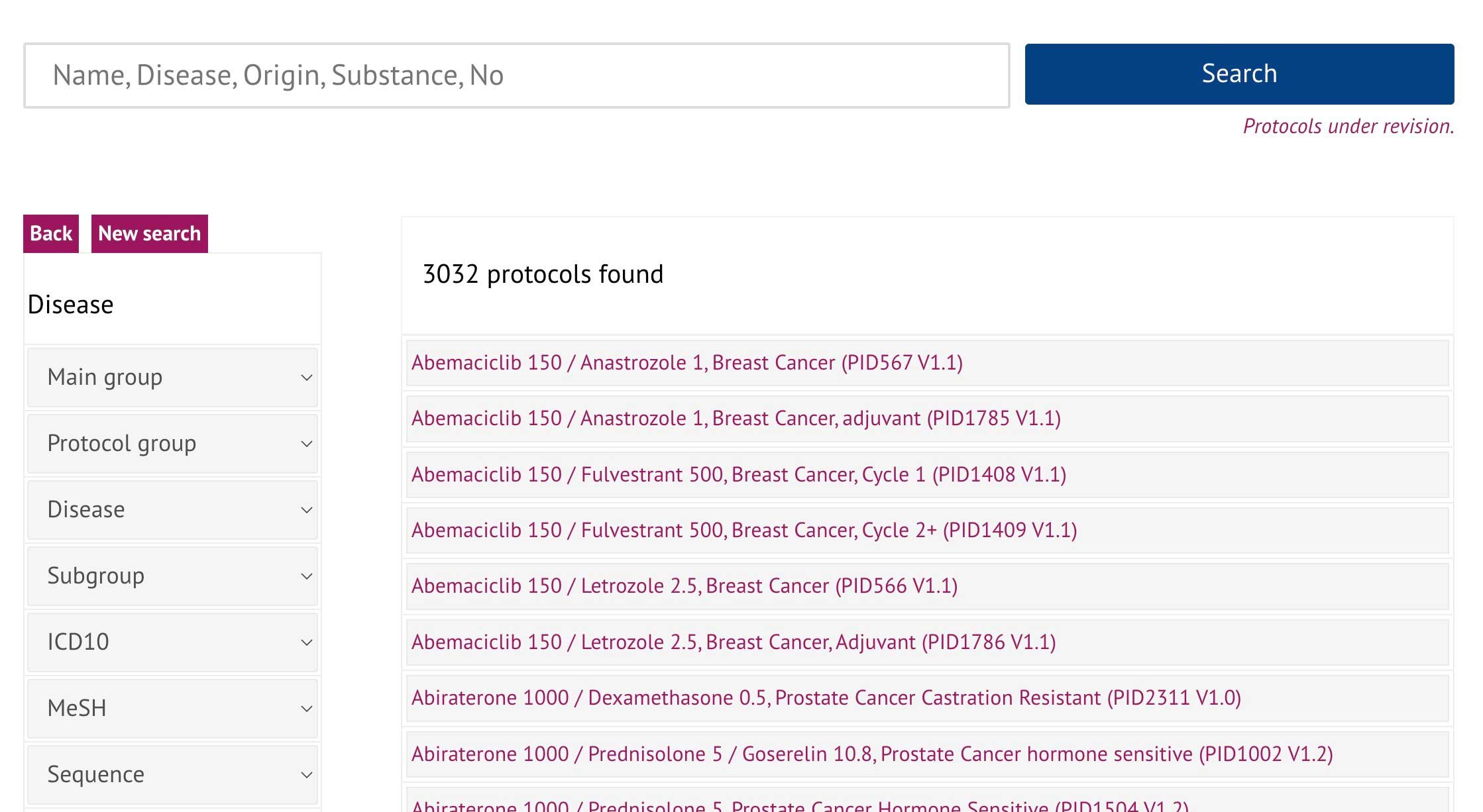
We provide a intelligent filter & search
for antineoplastic and supportive drugs
for diseases and subgroups
for disease categories for literature

To accommodate different professional requirements, our protocols are available through a tiered access model. Depending on the user's license level, the platform offers five distinct clinical views:
Mini: Available for non-registered visitors (Public access)
Basic: Enhanced data for registered users
Short to complete for professional subscribers

The protocols are assigned to clinically relevant main groups of diseases and therapeutic procedures.
Open our list of diseases and therapeutic procedures
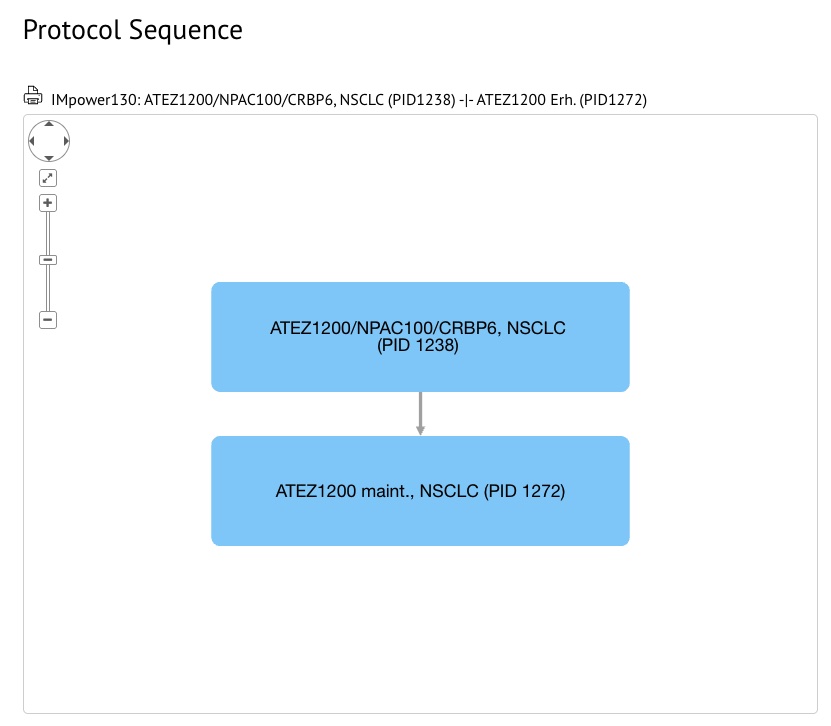
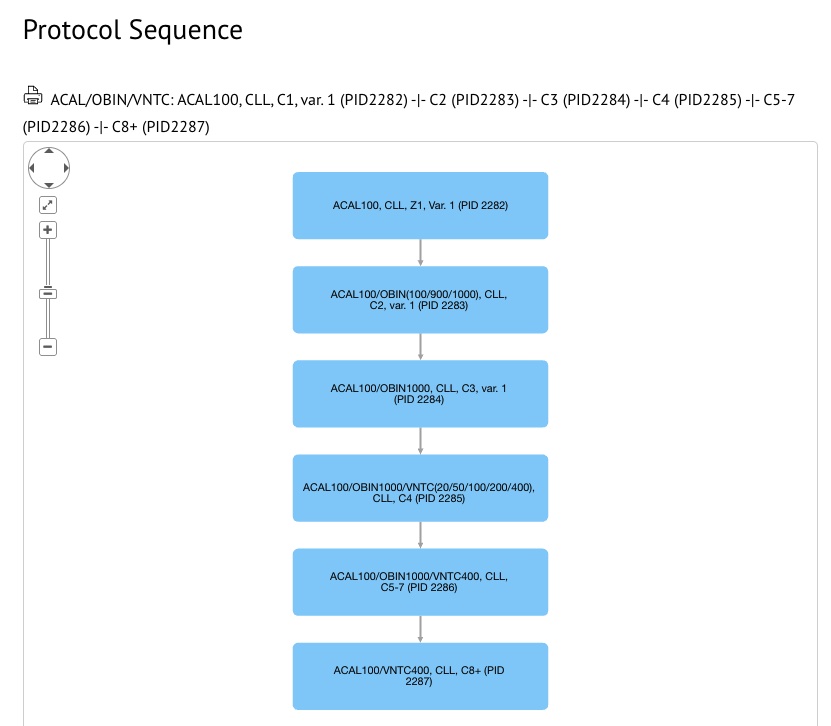
Protocol sequences